Most pharma R&D transformation efforts fail in the same way. Teams add artificial intelligence to old workflows. They digitize legacy processes. They sign new partnerships under old contracts. The work looks busy. The metrics barely move.
Meanwhile, Moderna compressed COVID-19 vaccine development from a decade to 11 months. Recursion runs millions of automated cell experiments weekly. Novartis unified 20 petabytes of R&D data across 2 million patient-years through its data42 platform. These are not technology stories. They are funding stories.
The companies winning the pharma R&D transformation race fund platforms first and programs second. The companies are losing funding programs and hope that platforms will emerge later. They never do.
This article gives you the rulebook: Five operating principles, three concrete shifts in portfolio management, and one 90-day starting plan. Use it to redirect capital, restructure governance, and shorten development timelines without waiting for the next budget cycle.
Why pharma R&D transformation fails at the funding stage
Drug development costs exceed $2 billion per approved drug. Timelines stretch beyond a decade. Phase I approval success rates sit below 10%. Adding research budget to that math does not fix it.
The structural problem is how money flows through pharma. Most companies still fund in annual cycles tied to programs. Each program rebuilds its own assays, its own manufacturing process, its own data pipeline. Capital spreads thin across one-off infrastructure that nobody else can reuse. Every program pays the setup tax twice: once when it starts, once when it tries to scale.
But atomic R&D inverts the flow. Companies fund the platform first. Assays, data backbones, manufacturing modules, and partner networks are paid for once and amortized across the portfolio. Programs become consumers of infrastructure, not builders of it. Unit economics flip: high upfront platform cost, low marginal cost per new asset.
Moderna runs 35+ mRNA programs from one platform. ADC Therapeutics relaunched a different approved drug within 18 months of a Phase 3 failure by reusing its antibody-drug conjugate platform.
The lesson from modern drug discovery: the platform is the asset. The molecule is a variable.
If your CFO measures success by program count and burn rate, your transformation is structurally blocked before it starts.
Five forces breaking the legacy drug development model
The legacy R&D model was built for one molecule, one process, one site, one trial. The life sciences industry no longer operates that way. Five forces have made the old design economically unviable, and none of them are reversing.
Cost without confidence.
Linear stage gates burn capital before signals get reliable. Decisions land too late. Discovery teams wait nine months for synthesis and assay results that platform-native companies deliver in weeks.
Modality complexity demands early integration.
mRNA, cell, and gene therapies require CMC, analytics, and clinical feasibility decisions from day zero. Late tech transfer turns interdependencies into 12-month delays and doubled budgets. Leading biologics teams that pull manufacturing into discovery cut tech transfer from 18-24 months to 6-12.
Data fragmentation is the hidden tax on speed.
Assays sit in one database. Clinical data lives in another. Batch records sit in a third. Researchers spend three weeks finding and reconciling data instead of running analyses. Without a governed data backbone, every new partner adds entropy.
Fixed protocols cannot keep pace with patients.
Site-centric trials enroll slowly. Amendment cycles add six months. The FDA now explicitly encourages adaptive designs and decentralized elements through 2019 and 2023 guidance documents. Sponsors that ignore the shift lose enrollment, lose access to diverse patients, and lose a competitive edge to faster operators.
Partnering is transactional when it should be strategic.
Most pharma companies still treat CROs and CDMOs as overflow. Each engagement becomes a custom project, with new SOWs and ad-hoc quality agreements. Strategic partners with pre-negotiated terms on board in three weeks. Transactional vendors take four months. External partnerships now account for over 50% of pipeline assets at top pharma companies. The model has already shifted across the industry. Many balance sheets and procurement processes have not.
These five forces will not reverse. The question is how fast you redesign around them.
The atomic rulebook for pharma R&D transformation
The atomic model breaks pharma R&D transformation into five operating rules. Each one is a funding decision before it is a process decision. Use the list as a filter on every initiative your team proposes this quarter.
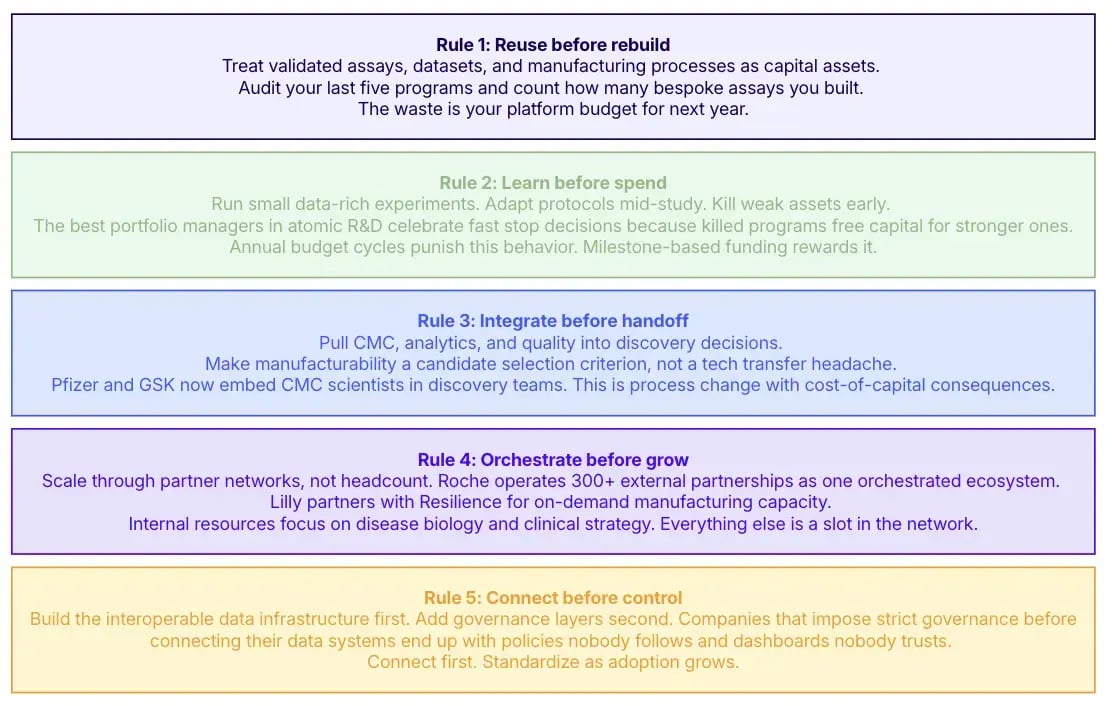
Exhibit 1: The rulebook for pharma R&D transformation
These rules apply equally to a 50-person biotech and a top-five pharma. The smaller company has fewer legacy systems to retire. The larger one has more capital to redirect. Both face the same atomic logic.
The honest test for any new R&D investment in your team: does it make a reusable capability stronger across the portfolio, or does it solve one program's local problem at the cost of building yet another silo?
Portfolio management built for atomic drug development
Portfolio management in the legacy model tracks programs. Atomic portfolio management tracks platforms, programs, and the link between them. Three concrete shifts follow.
Shift #1: separate platform funding from program funding
Treat platforms as multi-year investments with their own KPIs: assay reuse rate, data system adoption, partner cycle time. Programs draw from platforms and pay an internal "platform tax" of 2-3%. These funds shared infrastructure without net-new budget asks the CFO will reject.
Shift #2: add manufacturability and human-relevance to gate criteria
Most stage gates ask: "Does the molecule work in mice?" Atomic gates ask: "What is the human evidence? Can we make this at scale?" Add a six-criterion manufacturability scorecard at candidate nomination: titer, stability, aggregation risk, analytical tractability, raw material availability, scalability red flags. Investigational candidates failing the majority do not advance.
Shift #3: track learning velocity, not activity
Replace "programs in clinic" and "burn rate" with cycle time per design-make-test loop, decision latency at portfolio reviews, and right-first-time rate at tech transfer. Activity metrics reward motion. Velocity metrics reward learning, which is where competitive advantage actually compounds.
Johnson & Johnson built this kind of view through its Global Innovation Pillar. The pillar tracks hundreds of initiatives across regions and business units in real time. Stakeholders in R&D, supply chain, and digital functions see the same funnel: build an idea, fine-tune, learn from experiments, and deploy. When a project stalls, leadership identifies it the same week, not the next quarter.
Merz Therapeutics applied the same logic to technology scouting. The team consolidated clinical trial, patent, conference, and university research data into one platform. AI-enabled signal enrichment surfaces new developments automatically, supporting target identification and partnership decisions. The Business Development Board now receives curated opportunities, not raw lists. Portfolio decisions shifted from reactive to proactive.
Modern portfolio management for pharma R&D does one thing the old model could not. It surfaces the bottleneck while it is still cheap to fix.
Digital transformation that actually shortens drug development
Most digital transformation in the life sciences industry replaces paper with PDFs. That is digitization, not transformation. Real digital transformation in drug development changes which decisions get made, who makes them, and how fast.
Three plays separate the two.
Play #1: build a governed data backbone before adding AI on top
Novartis data42 unified 20 petabytes of R&D data across 2 million patient-years. The platform did not start with machine learning models.
It started with data contracts, lineage tracking, and FAIR standards. Once the backbone existed, AI models could be trained on trustworthy inputs. Companies that flip this order build expensive AI on noisy data and wonder why predictions fail.
Play #2: separate digital experiments from physical experiments
Recursion runs millions of perturbations per week through automated cell imaging and machine learning.
Pfizer used digital twins of bioreactors to optimize mRNA vaccine scale-up before committing physical batches. Insilico Medicine pairs generative AI with robotic labs to compress design-make-test cycles.
The pattern is consistent across these companies: artificial intelligence designs and predicts, physical labs validate. Each side scales independently.
Play #3: make CRO, and CDMO data flow through your systems, not around them
Manual data transfers (download, upload, reconcile) negate every productivity gain platform partners promise. Strategic partners write directly into your LIMS and EDC. Without this integration, you pay platform pricing for spreadsheet performance.
Industry surveys consistently report that 60% of AI partnerships stall because pharma data is unstructured, low quality, or trapped in legacy systems. Digital transformation that ignores the data layer underneath produces dashboards. Digital transformation that fixes the data layer first produces faster development timelines.
The test for any new technology investment in your team: does it shorten the next decision, or does it produce another report nobody reads?
How big Pharma shifts from builder to orchestrator
Big pharma is moving from vertical integrator to ecosystem orchestrator. The shift is not optional. Industry data shows external partnerships now account for over 70% of new molecular entity revenues. The companies that learn to orchestrate own the future. The companies that try to build everything internally lose the competitive edge to lighter, faster networks.
What orchestration looks like in practice:
Strategic core partners, not annual RFPs.
Select three to five partners (one CRO, one CDMO, one specialty vendor). Negotiate multi-year master agreements with shared KPIs and integrated data systems. Vendor onboarding drops from four months to three weeks. Procurement teams that re-RFP every year for marginal savings destroy this value. Executive sponsors must protect strategic relationships from short-term cost cutting.
Platform partners with defined slots in the value chain.
Treat external capabilities as standardized slots: high-throughput screening slot, AAV manufacturing slot, real-world evidence slot. Each slot has predefined data contracts, SLAs, and quality gates. Roche runs this model across 300+ partnerships. Lilly uses Resilience for on-demand biomanufacturing capacity. The slot concept is what makes external work feel like internal capacity.
Federated evidence networks.
Owkin connects hospitals through privacy-preserving AI to generate clinical research evidence without moving patient data. Roche integrates Flatiron Health's real-world oncology data into clinical trial decisions. These networks expand access to patients and data that no single sponsor could build alone, accelerating evidence generation for treatments in rare diseases and oncology.
The orchestration mindset changes how big pharma teams think about expertise. The question is no longer "Should we build this capability?" The question is "Where does this capability already exist at world-class quality, and how do we plug it in?"
This is also where commercial operations and commercial teams gain leverage. When R&D ships faster and cheaper, launch readiness, sales planning, and marketing handoffs compress with it. The value chain shortens end-to-end, from target identification through commercial launch.
Where to start: a 90-day implementation map
You cannot run all 13 atomic moves at once. Sequence matters. The right starting moves create the credibility and infrastructure that harder moves need.
Day 1 - 30: diagnose, do not transform
Run an atomic R&D readiness assessment across four dimensions:
-
decision authority (can program leads spend without committee approval?),
-
data maturity (can teams find data in minutes, not days?),
-
partnership capability (do you have pre-negotiated strategic agreements?), and
-
risk tolerance (are early kills celebrated as fast learning?).
Score each from 1 to 3. Your "1" scores are the first priority. Foundational gaps kill downstream moves.
Day 31 - 60: pick three moves and assign owners
Choose two quick wins and one foundational move. Quick wins build credibility. Foundational moves enable later transformation. A common starting trio:
- Compress design-make-test cycles in one drug discovery program (90-day pilot)
- Simplify protocols in your next clinical research design (run a patient burden audit)
- Begin a governed data backbone with two programs and one data type
Each move gets a named executive sponsor, an operational owner, and a 24-month budget protected from quarterly cuts. Project management discipline matters here. Without named accountability, transformation work loses to BAU work every time.
Day 61 - 90: instrument before you scale
Set up the dashboards before claiming results. Track cycle time, decision latency, rework rate, and platform reuse rate weekly. Without instrumentation, benefits stay anecdotal, and the budget gets cut at the next downturn.
The honest part: a data backbone breaks even at 18-24 months. Early CMC integration shows value 2-3 years out. Patient-centric protocols deliver 20-35% enrollment improvement in the first trial. Communicate timelines honestly. Underpromised quick wins beat overpromised transformations every time.
How ITONICS supports pharma R&D transformation
The atomic model demands one thing most pharma teams lack. It demands a system to connect insights, ideas, initiatives, partners, and platforms across the value chain in real time.
ITONICS provides that operational layer. Innovation and R&D teams use the platform to scout new technologies, evaluate startups, manage technology radars, and run portfolio management workflows that connect strategy to execution.
/Still%20images/Roadmap%20Mockups%202025/portfolio-link-projects-teams-and-milestones-2025.webp?width=2160&height=1350&name=portfolio-link-projects-teams-and-milestones-2025.webp)
Exhibit 2: A platform to prioritize processes with technology and project roadmaps
Johnson & Johnson, Roche, Merz Therapeutics, Novartis, and LivaNova use ITONICS to institutionalize the moves described in this article.
What teams gain in practice:
-
shared views of trends, technologies, competitors, and customer needs across all stakeholders;
-
structured workflows that turn raw signals into phase-gate-ready business cases with market evidence;
-
live governance views of status, risk, spend, and impact;
-
parent-child relationships that link platform investments to derivative programs, so platform ROI becomes visible at portfolio review.
The blueprint for Pharma R&D transformation already exists in this article. The system to deliver it is what separates teams that talk about transformation from teams that ship it.
FAQs on pharma R&D transformation
How long does a realistic pharma R&D transformation take?
Plan for 24-36 months to see structural results. Quick wins like patient-centric protocols deliver 20-35% enrollment improvement in the first trial.
A governed data backbone breaks even at 18-24 months. Early CMC integration shows attrition reduction at 2-3 years. The mistake is promising 12-month payback on infrastructure moves and getting cut at month 9 when the curve has not yet turned.
Where should we start if we have a limited budget?
Start with three moves chosen by readiness, not by ambition. Score your team across decision authority, data maturity, partnership capability, and risk tolerance from 1 to 3. Address your "1" scores first.
A typical starter pack: compress design-make-test cycles in one drug discovery program, simplify protocols on your next clinical trial, and pilot a data backbone on two programs with one data type. Total investment for the pilot stage is usually under $2 million.
Do we need new technology to begin?
No, but you need to stop layering new technology on broken processes. Fix decision authority and data flows first.
Industry surveys show 60% of AI partnerships stall because pharma data is unstructured, low quality, or locked in legacy systems. Spending on AI models before fixing the data layer wastes both budget and credibility.
How do we fund platforms when programs already consume the budget?
Three sources.
-
Reallocate 15% of IT spend from legacy systems.
-
Redirect duplicated data management labor across functions.
-
Implement a "platform tax" where new programs contribute 2-3% to shared infrastructure.
How does this apply to small biotech versus big pharma?
Small biotechs are already partially atomic by necessity. Their gap is usually in the project management discipline and data infrastructure as they scale past 5-10 programs.
Big pharma has the opposite problem: capital and expertise exist, but legacy systems and matrix governance block the redesign. The five rules apply to both. The starting point differs.
What is the single biggest failure mode?
Optional adoption. Squads launch, platforms deploy, partners sign on, but scientists keep their Excel workflows because "it is faster." Adoption stays below 30%. The transformation dies quietly.
The fix: make the old way harder than the new way. Retire legacy CRO processes by month 9. Freeze Excel tracking by month 12. Tie compensation to platform reuse and cycle time, not program count.




